(CartiHeal Demonstrates Agili-C™ Superiority to Microfracture and Debridement for the Treatment of Cartilage and Osteochondral Defects)
Bioventus, an American orthologics company, has exercised an option that it held to acquire Israeli medical device company CartiHeal. The price is $500 million with $360 million being paid out in front in cash.
Pursuant to its Option and Equity Purchase Agreement with CartiHeal Ltd., Bioventus made a $50 million escrow payment, sort of a deposit that ensures the company’s intention to go forward with the acquisition.
–
[embedded content]
In recent years Israel has become known as the startup state or Startup Nation. The country has had multi-billion dollar exits for numerous software and security companies. But Startup Nations is also known for its medtech startups. Israel has always been a center for medical research and innovation. Now put that together with its startup culture and you have medtech nation.
The examples are many. Israeli startup RealView Imaging offers innovations in medtech that use holographic imaging. Their HOLOSCOPE-I just got FDA approval. Append Medical, founded in 2017, develops the Appligator – a tool designed to reduce stroke risk in AF patients by excluding the LAA to prevent blood clot leakage with a minimally invasive transseptal intervention leaving no implant behind.
And some Israeli medtech startups are even using 3D printing to create body parts for transplant.
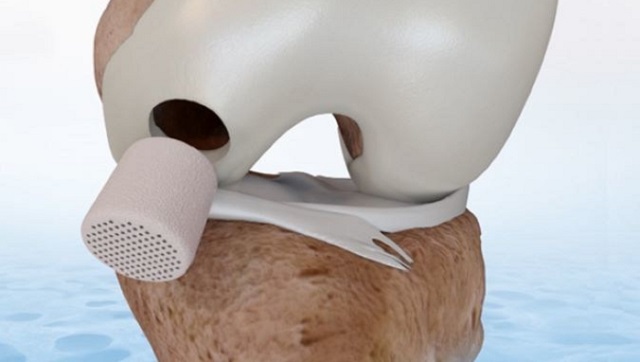
CartiHeal is a privately-held Israeli medical device company that develops proprietary implants for the treatment of cartilage and osteochondral defects in traumatic and osteoarthritic joints.
The Mayo Clinic explains that Osteochondritis dissecans (os-tee-o-kon-DRY-tis DIS-uh-kanz) is a joint condition in which bone underneath the cartilage of a joint dies due to lack of blood flow. This bone and cartilage can then break loose, causing pain and possibly hindering joint motion.
Osteochondritis dissecans occurs most often in children and adolescents. It can cause symptoms either after an injury to a joint or after several months of activity, especially high-impact activity such as jumping and running, that affects the joint. The condition occurs most commonly in the knee, but also occurs in elbows, ankles and other joints.
–
Bioventus Proceeds with Option Structure Agreement with CartiHealhttps://t.co/mMwHNB4bss #innovation #osteoarthritis #healthcare pic.twitter.com/wsnwjgIdnO
— Bioventus (@BioventusGlobal) August 30, 2021
CartiHeal offers a cell-free, off-the-shelf implant, CE marked for use in cartilage and osteochondral defects. Agili-C has been implanted in over 400 patients with cartilage lesions in the knee, ankle and great toe in a series of trials conducted in leading centers in Europe and Israel. In these trials, the implant was used to treat a broad spectrum of cartilage lesions, from single focal lesions to multiple and large defects in patients suffering from osteoarthritis.
In the United States the Agili-C implant is not available for sale – it is an investigational device limited for use in the IDE study.
“The robust data generated from the pivotal clinical trial, a randomized controlled trial with Agili-C, demonstrated superiority over surgical standard of care, microfracture and debridement, in KOOS overall compared to baseline. We believe this product could be a strong alternative for the approximately 650,000 US patients annually receiving microfracture or debridement along with other cartilage treatment options,” said Alessandra Pavesio, Senior Vice President and Chief Science Officer, Bioventus. “In combination with our HA products, Agili-C represents an exciting potential new offering for our portfolio designed to address the spectrum of osteoarthritis disease.”