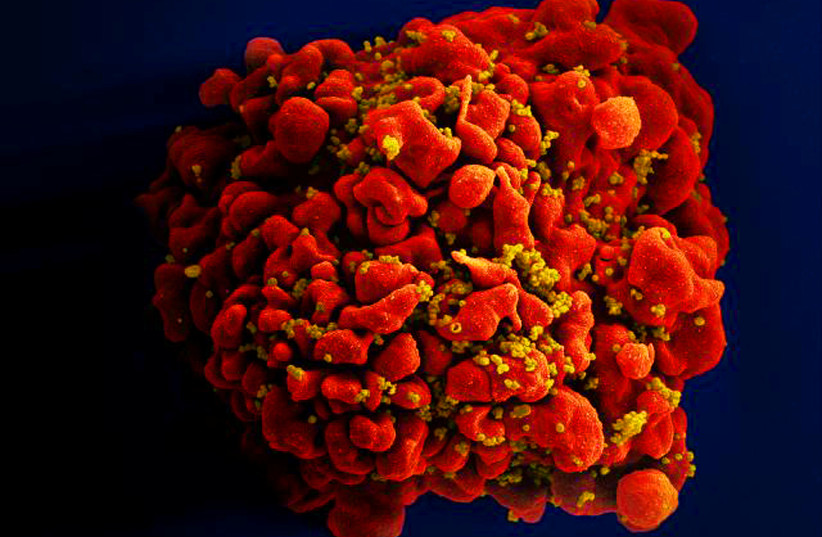
Scientists found that a new method to cure Human Immunodeficiency Virus (HIV) by transplanting stem cells resistant to HIV extracted from umbilical cord blood was successful.
According to a new peer-reviewed study that was published in Cell journal on Thursday, this process was used to successfully treat a mixed-race middle-aged woman with leukemia and HIV. Using stem cells from umbilical cord blood instead of from donors increases the potential to cure the disease through stem cell transplants in people of all racial backgrounds.
“The HIV epidemic is racially diverse, and it’s exceedingly rare for persons of color or diverse race to find a sufficiently matched, unrelated adult donor.”
Yvonne Bryson, UCLA
“The HIV epidemic is racially diverse, and it’s exceedingly rare for persons of color or diverse race to find a sufficiently matched, unrelated adult donor,” said Yvonne Bryson of the University of California, Los Angeles (UCLA), who co-led the study with Deborah Persaud, a pediatrician and infectious disease expert at the Johns Hopkins University School of Medicine. “Using cord blood cells broadens the opportunities for people of diverse ancestry who are living with HIV and require a transplant for other diseases to attain cures.”
Almost 38 million people globally live with HIV. Though antiviral treatments are effective, they need to be taken for the patient’s entire life. Several people have been cured of HIV since 2009. They all received stem cell transplants for cancer treatments and the donor cells came from compatible adults with two copies of the CCR5-delta32 mutation, which provides resistance to HIV by preventing the virus from entering and infecting cells.
Only about 1% of white people have two identical copies of the CCR5-delta32 mutation and other populations are even less likely to have them. This makes it difficult to transplant cells with the beneficial mutation into patients of color because stem cell transplants generally require a strong match between the donor and recipient.
Because of this, the scientists transplanted stem cells carrying CCR5-delta32 from banked umbilical cord blood to attempt to cure both the patient’s cancer and her HIV.
The transplant was completed in 2017 at Weill Cornell Medicine by a team of specialists led by Drs. Jingmei Hsu and Koen van Besien.
The case was part of the International Maternal Adolescent AIDS Clinical Trials (IMPAACT) Network, sponsored by the National Institutes of Health, and was co-endorsed by the Adult AIDS Clinical Trials Network (ACTG).
The blood cells from the umbilical cord blood were infused with stem cells from a relative of the patient in order to increase the likelihood that the procedure would succeed.
“The participant engrafted CBU cells from 107 weeks post-transplant were challenged with the two latent reservoir clones cultured from resting CD4+ T cells immediately before the haplo-cord transplant, along with laboratory strains HIV-1BAL (CCR5-tropic) and HIV-1NL43 (CXCR4-tropic) in a virus infectivity assay,” wrote the researchers.
“With cord blood, you may not have as many cells, and it takes a little longer for them to populate the body after they’re infused,” said Bryson. “Using a mixture of stem cells from a matched relative of the patient and cells from cord blood gives the cord blood cells a kick start.”
“Peripheral blood was collected before and following transplant—at study entry, Day 100 (week 15), week 27, 55, and approximately every six months thereafter (weeks 88, 107, 128, and 147) for immune profiling and quantitative markers of HIV-1 persistence,” the scientists wrote. “We collected bone marrow samples at weeks 15, 27, and 107 post-transplant to monitor AML status. We also analyzed remnant bone marrow cells for HIV-1 DNA concentrations. Quantifying the latent HIV-1 reservoir size was planned for pre-transplant and serially (weeks 15, 26, and 52) and then every six months following the transplant. However, we only collected blood samples for the latent replication-competent HIV-1 reservoir pre-transplant, at week 55 post-transplant, and again at week 2, 26 and 52 post-ATI.”
Following the procedure, the patient’s HIV and leukemia both went into remission that has lasted more than four years. Thirty-seven months after the transplant, she no longer needed to continue taking antiviral medication. She has now been HIV-negative for more than 30 months since she stopped taking the medication.
“Stem cell transplants with CCR5-delta32/32 cells offer a two-for-one cure for people living with HIV and blood cancers,” says Persaud. However, because of the invasiveness of the procedure, stem cell transplants (both with and without the mutation) are only considered for people who need a transplant for other reasons, and not for curing HIV in isolation; before a patient can undergo a stem cell transplant, they need to undergo chemotherapy or radiation therapy to destroy their existing immune system.
“This study is pointing to the really important role of having CCR5-delta32/32 cells as part of stem cell transplants for HIV patients, because all of the successful cures so far have been with this mutated cell population, and studies that transplanted new stem cells without this mutation have failed to cure HIV,” says Persaud. “If you’re going to perform a transplant as a cancer treatment for someone with HIV, your priority should be to look for cells that are CCR5-delta32/32 because then you can potentially achieve remission for both their cancer and HIV.”
Researchers highlight importance of increasing CCR5-delta32 screenings
The scientists said that more effort needs to go into screening stem cell donors and donations for the CCR5-delta32 mutation.
“With our protocol, we identified 300 cord blood units with this mutation so that if someone with HIV needed a transplant tomorrow, they would be available, but something needs to be done [on] an ongoing basis to search for these mutations, and support will be needed from communities and governments,” said Bryson.